| Generic Name: |
Pregabalin
|
| Therapeutic Category: |
Neuropathic Pain Agent
|
| Pharmacological Class: |
Alpha-2 delta ligand
|
| Composition: |
Pregalin 75
Each capsule contains Pregabalin 75 mg
Pregalin 150
Each capsule contains Pregabalin 75 mg
|
| Pregnancy Category: |
C
|
| Presentation: |
Pregalin-75
Available in the pack size as 10 tablets X 10 blisters
Pregalin-150
Available in the pack size as 10 tablets X 3 blisters
|
| Mechanism of Action: |
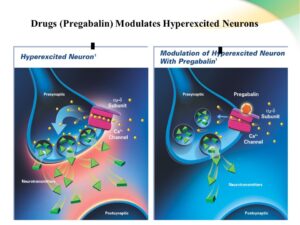 Pregabalin binds to ALPHA2-DELTA (α2-delta) subunit on voltage gated calcium channels in hyper-excited neurons. This inhibits influx of calcium and reduces release of glutamate, norepinephrine, substance P and other neurotransmitters. Pregabalin binds to ALPHA2-DELTA (α2-delta) subunit on voltage gated calcium channels in hyper-excited neurons. This inhibits influx of calcium and reduces release of glutamate, norepinephrine, substance P and other neurotransmitters.
Such decrease in neurotransmitter release from synapses in several neuronal tissues in the spinal cord and brain is likely to attenuate neuronal hyper-excitability and abnormal synchronization.
This may thus explain its anticonvulsant, analgesic, and anxiolytic activity
|
| Indications: |
- Neuropathic Pain Associated With Diabetic Peripheral Neuropathy (DPN)
- Post Herpetic Neuralgia* (PHN)
- Partial-Onset Seizures (epilepsy)
- Fibromyalgia
- Generalized Anxiety Disorder
|
| Dosage: |
Route of administration: Oral
- Neuropathic Pain Associated With Diabetic Peripheral Neuropathy:
150 mg daily in divided dose initially, increased to 300 mg daily in divided dose within 1 week based on efficacy and tolerability.
Maximum dose: 300 mg/day
- Partial-Onset Seizures:
75 mg twice daily initially, increased to 150 to 600 mg/day divided 2 or 3 times daily based on efficacy and tolerability
Maximum dose: 600 mg/day
- Post herpetic Neuralgia:
75 mg twice daily initially, increased to 150 mg twice daily within 1 week based on efficacy and tolerability.
Maximum dose: 600 mg/day
- Fibromyalgia:
75 mg twice daily initially, increased to 150 mg twice daily within 1 week based on efficacy and tolerability.
Maximum dose: 450 mg/day
|
| Pharmacokinetics: |
Absorption: Rapidly absorbed (greater than 90%)
Protein binding: Does not bind to plasma proteins
Metabolism: Negligible in humans
Route of elimination: Renal
Half-life: 6.3 – 11.5 hours
|
| Adverse Effects: |
- Very common (>10% of patients): dizziness, drowsiness
- Common (1–10% of patients): blurred vision, diplopia*, increased appetite and subsequent weight gain, euphoria*, confusion, vivid dreams, changes in libido (increase or decrease), irritability, ataxia*, attention changes, etc.
Withdrawal symptoms
Following abrupt or rapid discontinuation of Pregabalin, some people reported symptoms suggestive of physical dependence. Even people who have discontinued short term and or long term use of Pregabalin have experienced withdrawal symptoms, including insomnia, headache, nausea, anxiety, diarrhea, flu like symptoms, nervousness, major depression, pain, convulsions, hyperhidrosis and dizziness.
|
| Contraindications: |
PREGALIN is contraindicated in the following populations and situations:
- Hypersensitivity to Pregabalin or any other excipients of the formulation.
- Children, infants: The safety and efficacy of Pregalin have not been established in infants and children.
|
| Precautions: |
- Geriatric: The use of Pregalin in geriatric patients may require caution. Elderly patients are more likely to have reduced renal function and doses should be adjusted in the elderly patient based on the degree of renal impairment.
- Diabetes mellitus, heart failure
- Glaucoma, ocular disease
- Pregnancy, labor, obstetric delivery
|
| Drug Interactions: |
- Potential pharmacological interactions with opioids, benzodiazepines, barbiturates, ethanol (alcohol), and other drugs that depress the central nervous system.
- ACE inhibitors may enhance the adverse/toxic effect of Pregalin.
- Pregalin may enhance the fluid-retaining effect of anti-diabetic agents (Thiazolidinedione)
|